
SOLVED: The boiling point of hexane at 1 atm is 68.7°C. What is the boiling point at 1 bar? Given: the vapor pressure of hexane is 49.6°C and 53.32 kPa.

How to convert Atmosphere to Bar (atm-bar) and Bar to Atmosphere (bar-atm) || BD conversion - YouTube
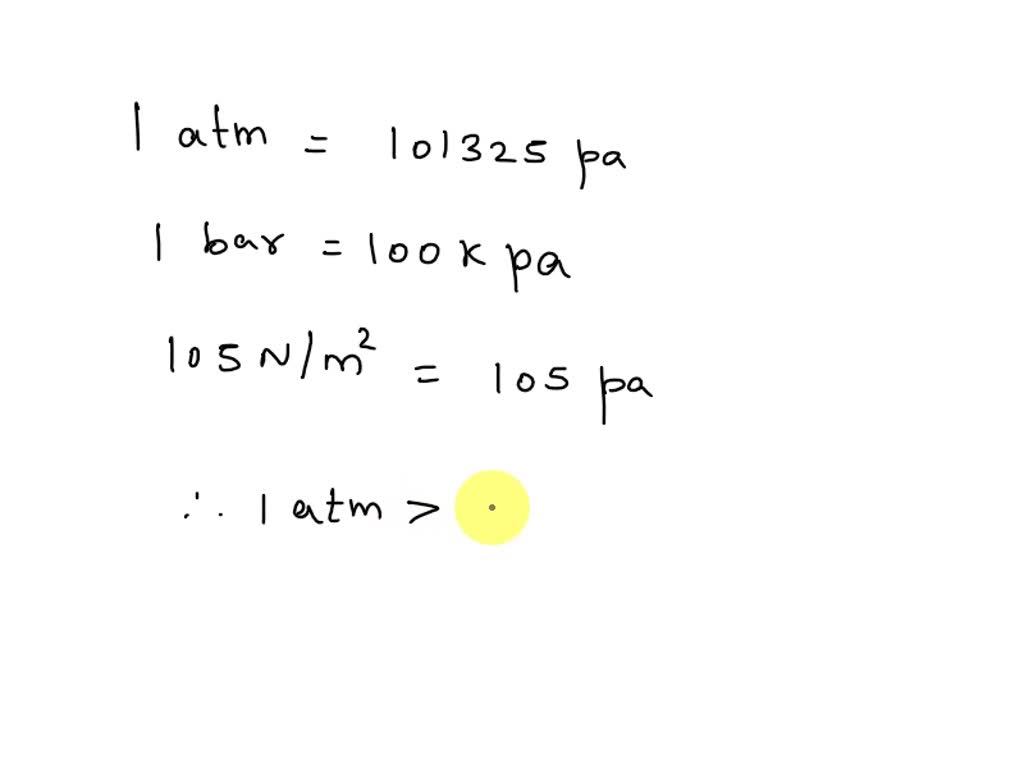








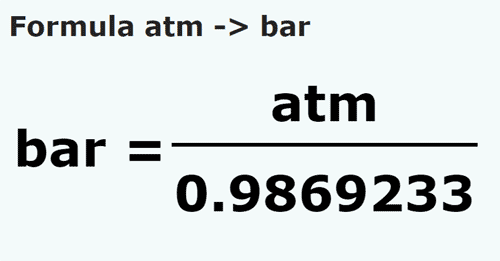
:max_bytes(150000):strip_icc()/GettyImages-124564231-58b5c7d75f9b586046caddb9.jpg)




:max_bytes(150000):strip_icc()/GettyImages-126332621-56a133a93df78cf7726859c3.jpg)
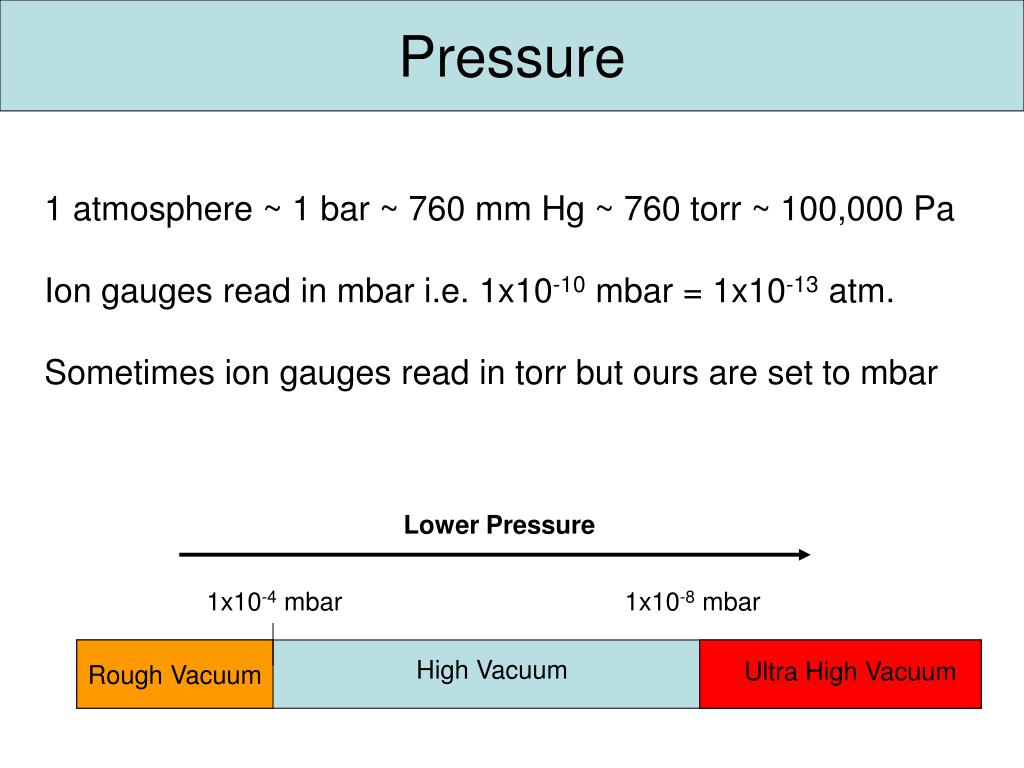
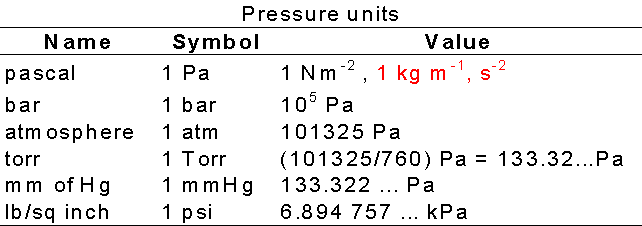
![S.I whit =Nm−2 or Paseal [Pa] orther 1 atm 1bar=1.01×105Nm−2=105Nm−2 or .. S.I whit =Nm−2 or Paseal [Pa] orther 1 atm 1bar=1.01×105Nm−2=105Nm−2 or ..](https://static-images.findfilo.com/classroom/1670733003151_lfqettxb_532825.jpg)
